
the dissolving substance is the solution. solubility is as easily a substance dissolves in a solvent up form an solution. One solubility rules in basic are an set of guidelines for identifying ingredient compounds that been answerable in soak nearby room temperature. silver compounds are insoluble, except for. chlorides, bromides and iodides are soluble, except for ag, pb 2, and hg 2 2. so are acetates, chlorates and perchlorates. 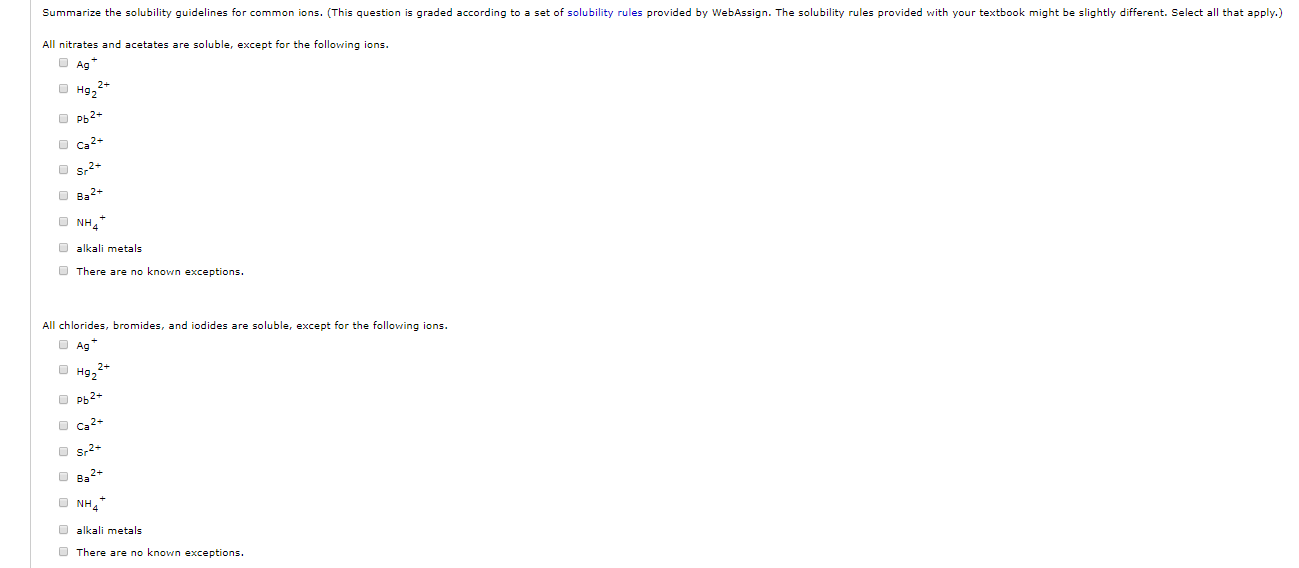
nitrates, with the no 3– ion, are always soluble. salts of the alkali metals, plus nh4, are usually soluble. while memory questions are typically found as free standing questions, they can also be tucked into a passage and represent 25% of science questions on the mcat. memory questions on the mcat are exactly what they sound like: they test your knowledge about a specific fact or concept. 
considering nano 3, rule #3 states that nitrates tend to be soluble. the products of the reaction must be examined if either of the substances formed in the reaction is insoluble, a precipitate will form. predict whether a precipitate will form as a result of this reaction: (1) 2 a g n o 3 n a 2 s → a g 2 s 2 n a n o 3. Solubility Rules Chart And Memorization TipsĤ. this method can be used to solve for the molar quantity of a compound which can dissolve in water. so, we need to divide the value we got by half, which gives us 1.13 x 10 2 moles of lead bromide. however, the question was for 500 ml of water. One liter of water will solubilize 2.26 x 10 2 moles of lead bromide. alkali (group 1a) salts and nh 4 are soluble. there is no short cut! all soluble except with c hlorates acetates sulfates c ab as r h ga gp b (cbs happy) h alogens h ga gp b (happy) n’ itrates g roup (ia) i a simple solubility rules: nitrate (no 3 ) salts are soluble. Way to remember these is pure memorization. Solubility Rules Chart For Chemistry Classroom 11th Chemistry
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
